Mario Hofmann of National Cheng Kung University shows the example set up of electrochemical synthesis.
Image: Mario Hofmann/IOP Publishing
Graphene has been affectionately coined the “wonder material” due to its strength, flexibility, and conductive properties. The theoretical applications for graphene have included the five-second phone charge, chemical sensors, a way to soak up environmentally harmful radioactive waste, and even the potential to improve your tennis game. While everyone has big expectations for the wonder material, it’s still struggling to find its place in the world of materials science.
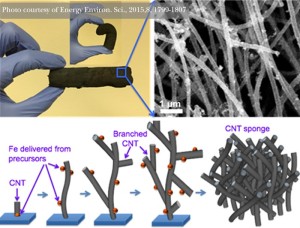
However, a team of researchers may have found a way to expand graphene’s potential and make it more applicable to tangible devices and applications. Through a simple electrochemical approach, researchers have been able to alter graphene’s electrical and mechanical properties.
Technically, the researchers have created a defect in graphene that can make the material more useful in a variety of applications. Through electrochemical synthesis, the team was able to break graphite flakes into graphene layers of various size depending on the level of voltage used.
The different levels of voltage not only changed the material’s thickness, it also altered the flake area and number of defects. With the alternation of these three properties, the researchers were able to change how the material acts in different functions.
“Whilst electrochemistry has been around for a long time it is a powerful tool for nanotechnology because it’s so finely tuneable.” said Mario Hofmann, a researcher at National Cheng Kung University in Taiwan, in a press release. “In graphene production we can really take advantage of this control to produce defects.”
The defected graphene shows promising potential for polymer fillers and battery electrodes. Researchers also believe that by revealing and utilizing the natural defects in graphene, strides could be made in biomedical technology such as drug delivery systems.