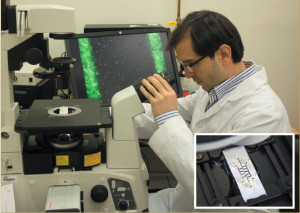
ECS student member Alireza Mahdavifar observes live bacteria moving inside the microfluidic channel.
Image: Georgia Tech/The Poultry Site
Along with a team of researchers out of Georgia Tech, ECS student member Alireza Mahdavifar has designed and fabricated the prototype of a microfluidic device that exploits cell movement to separate live and dead bacteria during food processing.
The research, entitled “A Nitrocellulose-Based Microfluidic Device for Generation of Concentration Gradients and Study of Bacterial Chemotaxis,” has been recently published in the Journal of The Electrochemical Society.
The new development consists of a microfluidic device that exploits cell movement to separate live and dead bacterial during food processing. The device is novel due to the fact that while screening for foodborne pathogens, it can be difficult to distinguish between viable and non-viable bacteria. Mahdavifar and the team out of Georgia Tech responded to this issue by creating a device that can separate live cells from dead ones for real-time pathogen detection.